Purpose
Intermediate-risk prostate cancer patients represent the largest of the risk groups and comprise a heterogeneous population of patients with variable prognoses [1]. According to National Comprehensive Cancer Network Criteria (http://www.nccn.org/) intermediate-risk prostate cancer patients were defined as intermediate-risk if they fulfilled at least one of the following criteria: prostate-specific antigen (PSA) level 10-20 ng/ml, and/or Gleason score = 7, and/or clinical stage T2b or T2c.
Patients within the intermediate-risk category experienced a significant biochemical recurrence (at least 30%) following treatment with radical prostatectomy or external beam radiation therapy (EBRT) [2,3]. These reports also described heterogeneity of both biochemical and clinical recurrence rates in the intermediate-risk prostate cancer patients. Based on this heterogeneity of intermediate-risk prostate cancer, a new classification subdividing patients with intermediate-risk prostate cancer into “favorable” and “unfavorable” subgroups has been proposed:
favorable intermediate-risk (FIR): one IR factor with Gleason score 3 + 4,
unfavorable intermediate-risk (UIR): Gleason 4 + 3 = 7 or > 1 intermediate-risk factors (cT2b, cT2c, PSA 10-20, Gleason 3 + 4 = 7).
The probability for biochemical recurrence in UIR patients is significantly higher than that in FIR patients following treatments with radical prostatectomy or EBRT of 81 Gy [4,5]. We have previously obtained excellent results in terms of biochemical failure-free survival (BFFS) and local control in high-risk and very high-risk cancer, including cases with nodal metastasis, by high-dose (biologically effective dose – BED > 220 Gy) radiotherapy by low-dose-rate (LDR) brachytherapy in combination with EBRT: the data showed a 5-year actual BFFS rate of 95.2% [6].
In our present study on the efficacy and toxicity of LDR brachytherapy-based radiotherapy in our institution, we report clinical outcomes for intermediate-risk prostate cancer patients, including both favorable and unfavorable groups, treated with LDR brachytherapy-based radiotherapy with a BED ≥ 200 Gy.
Material and methods
Patients
This retrospective and observational study was conducted in accordance with the Helsinki Declaration. This study has been approved and monitored by our institutional ethics committee (R-2019-120).
From 2005 to 2016, a total of 397 patients with intermediate-risk prostate cancer were treated by LDR-based radiotherapy with a BED > 200 Gy.
The patients were classified according to the National Comprehensive Cancer Network Criteria (http://www.nccn.org/): briefly, patients were defined as intermediate-risk if they fulfilled at least one of the following criteria: prostate-specific antigen (PSA) 10-20 ng/ml, and/or Gleason score = 7, and/or clinical stage T2b or T2c.
The intermediate-risk patients in the present study included any type of intermediate-risk patient: both favorable and unfavorable intermediate-risk patients.
Staging
Clinical T stage was determined by a combination of magnetic resonance imaging (MRI) and digital examination. All patients had bone scans and computed tomography (CT) of the pelvis to check for the presence of bone metastasis and lymph node metastasis.
Clinical characteristics
The clinical characteristics of the patients (PSA, Gleason score clinical T stage) are shown in Table 1. The distribution of the number of intermediate-risk factors is shown in Table 2. A total of 145 cases were diagnosed as having primary Gleason pattern 4: Gleason score 4 + 3 (36.5%).
Table 1
Patient and disease characteristics of the 397 intermediate-risk cases are shown
Treatment
A total of 397 patients with intermediate-risk prostate cancer were treated by LDR-based radiotherapy with a BED ≥ 200 Gy. Treatment consisted of LDR brachytherapy alone (177 cases) or LDR and EBRT (220 cases). Short-term (3-6 months) androgen deprivation therapy (ADT) was used in 186 patients (46.9%).
Androgen deprivation therapy consisted of gonadotropin-releasing hormone agonist injection and anti-androgen. LDR brachytherapy implantation in the prostate was conducted with 125I seeds using real-time ultrasound guided technique [7]. Radioactive seeds were deposited into the prostate using a Mick applicator (Mick Radio-Nuclear Instruments, Ind., Mount Vernon, NY).
The prescription dose of seed implantation was set at 144 Gy for LDR monotherapy and 110 Gy for LDR combined with EBRT. In order to achieve high dose seed implantation, we set D90 at 190 Gy for LDR monotherapy and D90 at 135-140 Gy for LDR and EBRT combination therapy upon seed implantation. To achieve this goal, we intentionally made a high dose cloud (dose areas of 240 Gy for LDR monotherapy and 160 Gy for LDR and EBRT combination therapy) intentionally along the periphery (bilateral wall to anterior wall of the prostate) away from the urethra and rectum: the detailed report on this high dose seed implantation technique is now under preparation separately. For this method, we routinely used seed activity at 11.0 MBq.
In both LDR monotherapy and LDR combined with EBRT, the radiation dose was set to achieve a total BED of 200 Gy: Post-implant dosimetry with CT and MRI guidance was carried out one month after seed implantation.
For LDR monotherapy, D90 of the prostate was set over 190 Gy so that the post-implant BED would be higher than 200 Gy.
For combination therapy with LDR and EBRT, supplemental EBRT was delivered four to eight weeks after seed implantation. EBRT consisted of a median dose of 45 Gy given in 1.8 Gy fractions via a three-dimensional conformal technique.
In each case, the BED was calculated from the prostate D90 of the LDR and EBRT dose using the formula described previously [8]: the EBRT dose was determined so that the total BED would be higher than 200 Gy as long as UD30 and R100 were tolerable. EBRT fields included prostate and seminal vesicles only with a margin. Clinical target volume (CTV) was designed as the entire prostate and seminal vesicle. Planning target volume (PTV) included CTV-block with a 15 mm margin except at the prostatorectal interface, where a 7-10 mm was used.
Trend change of treatment modalities
To see our treatment trends during our 15-year LDR experience, we examined the modality ratio among LDR monotherapy and combination therapy with LDR and EBRT for 3 years.
Follow-up and statistical analysis
Scheduled follow-ups were done by PSA blood test and physical examination every three months for the first two years, followed by every six months thereafter. The follow-up duration was calculated from the time of the LDR for LDR monotherapy, and from the end of the supplemental EBRT for combination therapy. These patients had a minimum follow-up time of two years: the median follow-up was 72 months (range 29-165 months).
Actuarial survival curves were calculated by the Kaplan-Meier method to determine BFFS, freedom from clinical failure (FFCF) survival, cause-specific survival (CSS) and overall survival (OS). Biochemical failure was defined according to the Phoenix Definition [8]. The criterion for biochemical failure with subsequent PSA decrease to < 0.5 ng/ml without intervention was categorized as a benign bounce and was excluded from the biochemical failure group. Upon a true biochemical failure, we performed CT, MRI, bone scan and rectal digital examination to see whether biochemical failure was caused by distant metastasis or local failure.
Biochemical failure-free survival was calculated for all living patients and reflected biochemical failures. FFCF survival rate was calculated for all living patients and reflected clinical failure events (local, regional and distant failure). CSS reflected prostate cancer-specific death. OS reflected all deaths, cancer related or unrelated.
Toxicity
Acute toxicity was defined when symptoms developed within the first year after seed implantation. Late toxicity was defined when any kind of symptom developed after one year or when any symptom occurred within the first year and persisted for more than one year. Toxicity was recorded by the Common Terminology Criteria for Adverse Events version 4.0.
Results
Trend change in treatment modalities
The trend changes of our treatment modalities for every three years of our 15-year LDR experience are shown in Figure 1.
Fig. 1
Trend change of treatment modalities in our 15-year LDR experience. Modality ratio among LDR monotherapy (white bar) and combination therapy with LDR and EBRT (gray bar) for 3 years are shown
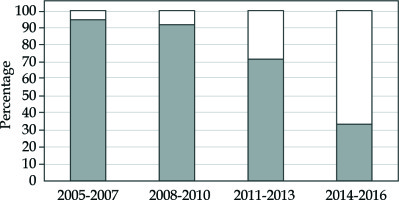
During the early period, we used combination therapy with LDR and EBRT with or without ADT for intermediate-risk patients. The reason for using LDR and EBRT combination therapy was to deliver high BED > 200 Gy for intermediate-risk patients. In those days, we did not have sufficient technique to deliver high BED of 200 Gy without using EBRT. During the early period, we also used ADT particularly for UIR cases or cases with prostate volume ≥ 40 ml. In 2012, we stopped using ADT in any type of intermediate-risk patients.
Our current treatment policy is that BED is the most crucial factor for local control of intermediate-risk prostate cancer. Therefore, we do not use EBRT or ADT in most cases of intermediate-risk prostate cancer including UIR cases at present.
Dosimetric parameters and BED
Dosimetric parameters of LDR monotherapy (N = 177) based on the data at one month after LDR are shown in Table 3.
Table 3
Dosimetric parameters of seed implantation at one month and calculated BED in the 177 cases treated by LDR monotherapy is shown
The average D90 and BED of LDR monotherapy were 192.5 Gy and 203 Gy, respectively.
Dosimetric parameters of combination therapy with LDR and EBRT (N = 220) based on the data at one month after LDR are shown in Table 4.
Table 4
Dosimetric parameters of seed implantation at one month and calculated total BED with LDR and EBRT in the 220 cases treated by combination therapy with LDR and EBRT are shown
The average D90 of LDR and total BED of the combination therapy were 133.9 Gy and 220.3 Gy, respectively.
Efficacy of the treatment
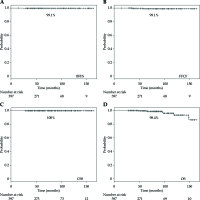
Three patients developed biochemical failure, thus providing a 7-year actual biochemical failure-free survival (BFFS) rate of 99.1% (Figure 2). Biochemical failure was observed exclusively in cases with distant metastasis: two cases with lymph node metastasis and one case with bone metastasis, thus yielding a 7-year freedom from clinical failure (FFCF) rate of 99.1% (Figure 2). We observed eight deaths, but there was no death from prostate cancer, thus yielding a 7-year cause-specific survival (CSS) rate of 100%, and an OS rate of 98.4% (Figure 2).
The number of intermediate-risk features of the patients who developed biochemical failure
We analyzed the number of intermediate-risk features of the three patients who developed biochemical failure.
Case 1 with two intermediate-risk features: clinical failure with bone metastasis. Pretreatment PSA = 11.0 ng/ml, T2c, Gleason score = 3 + 3. Case 2 with one intermediate-risk feature: clinical failure with lymph-node metastasis. PSA = 7.8 ng/ml, T2b, Gleason score = 3 + 3. Case 3 with three intermediate-risk features: clinical failure with lymph-node metastasis. PSA = 11.2 ng/ml, T2b, Gleason score = 3 + 4. Thus, occult metastasis existed regardless of the number of intermediate-risk patients.
Toxicity
Acute grade 2 genitourinary (GU) and gastrointestinal (GI) toxicity was experienced by 44 patients (11.1%) and two patients (0.5%), respectively. Late grade 2 GU and GI toxicity was experienced by 48 patients (12.0%) and five patients (1.3%), respectively. Grade 3 GU toxicity was not observed.
Grade 3 GI toxicity was observed in one patient (0.3%): R100 of this case at one month after seed implantation was 1.8 cc. Therefore, we reduced the EBRT dose down to 30.6 Gy. However, the patient experienced grade 3 rectal bleeding and received a blood transfusion. The patient recovered well without recurrent rectal bleeding after that.
This patient was an early-experience case (case number 20 of the 397 cases) treated by combination therapy with LDR and EBRT. The patient is alive and has since been free from rectal bleeding.
None of the patients experienced urethral stricture, TUR-P (transurethral resection of prostate), or recto-urethral fistula.
Discussion
In the present study, we demonstrated that a series of intermediate-risk prostate cancer patients in our institution obtained excellent recurrence-free survival either by LDR monotherapy or by combination therapy and LDR.
Notably, our intermediate-risk patients included a significant number of UIR patients. Overall, only three patients (0.8%) experienced biochemical failure (BFFS rate of 99.1% at seven years). Biochemical failure was observed exclusively in cases with distant metastasis (lymph node metastasis in two cases and bone metastasis in one case) (FFCF rate of 99.1% at seven years).
Radical prostatectomy is one of the standard treatment modalities for intermediate-risk prostate cancer. A group at Johns Hopkins University reported on BFFS in a cohort of 4,164 intermediate-risk patients. The results showed that 5-year BFFS differed significantly between FIR patients and UIR patients: For patients with one intermediate-risk factor, the 5-year BFFS was 83.0%, compared with 64.3% for men with two risk factors and 45.9% for those with three risk factors [4]. Similar differences in BFFS between FIR patients and UIR patients have been reported for EBRT of 81 Gy [5]. In their report, the estimated 8-year BFFS rates are 86.1% and 71.1% in FIR patients and UIR patients, respectively. The estimated 8-year local failure rates are 9.1% and 12.4% for FIR patients and UIR patients, respectively. The 8-year prostate cancer-specific mortality rates are 2.0% and 4.2% in FIR patients and UIR patients, respectively.
Grimm et al. conducted a large-scale comprehensive review of the literature comparing risk-stratified patients by treatment option and with long-term follow-up [9]. The outcome for intermediate-risk patients varied significantly among the treatment options. At 8 years, the estimated median BFFS rates for intermediate-risk patients were 90%, 85%, 70% and 70% among combination therapy with LDR and EBRT, LDR monotherapy, radical prostatectomy, and EBRT. In each treatment modality, BFFS differed substantially among the reports or institutions. There may be several reasons for those differences: 1) technical reasons, 2) selection bias of intermediate-risk patients.
Use of prostate brachytherapy provides the advantage of safely delivering a high biologically effective dose (BED) to the prostate [8,10,11].
It has been debated whether there is a need for supplemental EBRT when applying LDR-based radiotherapy in intermediate-risk cancer patients [12,13]. In those debates, there is a clear agreement that dose escalation is a key to improving outcomes for intermediate-risk patients, including UIR patients [12,13].
The advantage of combination therapy with LDR brachytherapy and EBRT has been recently confirmed by the ASCENDE randomized trial [14].
Indeed, we have previously shown that high-dose (BED > 220 Gy) radiotherapy by LDR in combination with EBRT leads to excellent BFFS and local control in high-risk and very high-risk cancer [6].
A crucial question remains how high BED is required for intermediate-risk patients including UIR cases. Based on the previous review [12], we suggest that a BED of 200 Gy is high enough for intermediate-risk patients, including UIR cases.
The present study on intermediate-risk cancer included a significant number of UIR patients. We did not conduct a statistical analysis on the BFFS rate among FIR and UIR patients in this study because biochemical failure was observed in only three cases. Therefore, the present study may at least prove the efficacy of outcome by our LDR-based radiotherapy for intermediate-risk prostate cancer patients, including UIR cases.
Furthermore, our series of patients did not experience any local recurrence within the present follow-up period, although much longer follow-up is necessary to exclude this possibility.
The present data may suggest that a BED of 200 Gy is high enough for eradicating both FIR and UIR prostate cancer. Our treatment trend change in intermediate-risk prostate cancer patients has moved forward to LDR monotherapy without ADT: This trend became evident in the last five years.
The background for this trend change is based on the technical advances in delivering a BED of 200 Gy safely by LDR monotherapy and accurate seed implantation for large volume prostate regardless of pubic arch interference (unpublished data).
Our study limitations included: 1) the short period of follow-up; 2) the retrospective character of this study. Even considering these shortcomings, this study suggests that LDR-based radiotherapy with a BED ≥ 200 Gy may result in good BFFS in intermediate-risk prostate cancer patients, including UIR.
Conclusions
This study shows an excellent outcome for intermediate-risk prostate cancer patients treated with BED ≥ 200 Gy radiotherapy using LDR brachytherapy. LDR alone achieving a BED > 200 Gy may be considered an optimal treatment in both favorable and unfavorable intermediate-risk prostate cancer patients, although a longer follow-up is mandatory.