Shivering is a common unpleasant perioperative complication of neuraxial anaesthesia. It has been reported in 40–70% of regionally anaesthetized patients. Shivering is a physiological compensatory response to core hypothermia due to redistribution of heat as a result of vasodilatation from chemical sympathectomy of spinal anaesthesia, exposure to a cool environment, infusion of unwarmed fluids and evaporation from exposed surfaces [1].
During neuraxial anaesthesia, not only is core heat redistributed from the trunk (below the sympathectomy level) to the periphery, but also the thermoregulatory system is significantly impaired due to the inhibited tonic vasoconstriction [2]. Moreover, the shivering threshold is reduced by about 0.5°C during neuraxial anaesthesia [3]. The exposure of the thermosensitive structures within the spinal cord to a cold local anaesthetic is another contributing factor [4].
Post-spinal shivering presents with different degrees ranging from skin eruption in the mild form up to generalized skeletal muscle contraction in the severe form that interferes with oxygen saturation, pulse rate, blood pressure and electrocardiographic monitoring [3].
Shivering increases the metabolic rate, the oxygen consumption and carbon dioxide production with lactic acidosis, which is hazardous in patients with poor cardiac and/or respiratory reserves. It also increases intraocular and intracranial pressures and leads to hypertension, tachycardia, patient discomfort, increased wound pain, delayed wound healing, and delayed discharge from the post-anaesthetic care unit (PACU) [1, 5]. That is why its primary prevention and prompt control are warranted.
Nonpharmacological measures such as maintaining perioperative normothermia, infusion of warm fluids, using warm covers and warm sheets have been recommended to prevent shivering [1]. Pharmacological agents such as clonidine (α2 agonist) and tramadol (μ-opioid agonist) possess anti-shivering properties [6]. Also, pregabalin has been reported to exert anti-shivering effects [7]. Perioperative pain and stress may promote non-thermoregulatory shivering and increase the thermoregulatory set-point [8].
Considering the spinal anaesthesia, the continuous uterine irrigation with fluids during hysteroscopic procedures and the higher incidence of shivering in young females together with known analgesic and anxiolytic properties of tramadol, clonidine and pregabalin, we aimed to compare the efficacy and safety of these drugs as a prophylaxis against post-spinal shivering [9].
METHODS
This randomized prospective triple-blinded placebo-controlled trial was conducted at Menoufia University Hospitals between September 2017 and November 2018 after approval from the local ethics and research committee. Informed written consent was obtained from each patient as per the ethical protocols and guidelines. The trial was registered at www.pactr.org (PACTR201710002423425). The study was conducted in concordance with the Consolidated Standards of Reporting Trials (CONSORT) guidelines.
We studied 120 American Society of Anesthesiologists (ASA) physical status I or II female patients who were scheduled for operative hysteroscopic procedures with an anticipated duration of about 60 minutes under spinal anaesthesia. The participants were 18-60 years old at the time of enrolment in the study. Patients who were feverish or hypothermic before the procedure were excluded. Patients with a history of hypertension, cardiovascular, psychiatric, neurological, endocrine, chest disease, and/or severe renal or hepatic dysfunction were excluded. In addition, we did not include obese patients (BMI ≥ 30 kg m−2), those who needed intraoperative blood transfusion and those with known allergy to clonidine, pregabalin, tramadol or any of this study’s drugs. Also, participants who had a history of drug or alcohol abuse and/or those who were on β blockers, vasodilators, gonadotropin, tramadol, clonidine or pregabalin before the surgery were excluded. Any contraindication to neuraxial block was an exclusion criterion.
None of the patients, the data collector or the outcome assessors was aware of the given premedication. The participants were randomly allocated to one of four groups of 30 patients each using a randomization table that was created by a computer software program. The randomization list was concealed in sealed opaque envelopes. Meanwhile, the entire drug preparation and administration process was performed by an anaesthesiologist who was not involved in the study.
Before administration of the oral premedication, each patient’s baseline heart rate, mean systemic arterial pressure, pulse oximetry, forehead skin temperature, tympanic membrane temperature and sedation score were measured. Ninety minutes before administering the intrathecal anaesthesia, group C patients (n = 30) received a tablet containing clonidine (0.2 mg) and a multivitamin capsule, while group P patients (n = 30) received a pregabalin (150 mg) capsule and a multivitamin tablet, group T (n = 30) patients received a tramadol (100 mg) capsule and a multivitamin tablet, and group O received a multivitamin tablet and a multivitamin capsule. All oral premedication drugs were given with a sip of water.
All the anaesthetic and surgical techniques were standardized. On arrival at the operating room, continuous electrocardiogram, non-invasive blood pressure and pulse oximetry monitors were applied. The body temperature was monitored at the forehead skin and the tympanic membrane every 5 minutes intraoperatively and every 30 minutes postoperatively. Baseline readings of all the parameters were recorded.
An 18-gauge cannula was inserted in a peripheral vein and a warmed (37°C) lactated Ringer solution infusion (7 mL kg−1 h−1) was started. Granisetron (1 mg) was administered intravenously as a prophylaxis against nausea and vomiting. Thereafter the patient was supported in the sitting position. Intrathecal anaesthetic (room temperature hyperbaric bupivacaine 0.5% (12.5 mg) + fentanyl 25 μg (0.5 mL) was given under complete aseptic technique using warm Betadine (37ºC) then the patient was returned to the supine position. After ascertaining adequacy of the block, the patient was positioned in the lithotomy position. The operating theatre was conditioned to maintain constant humidity of 65% and ambient temperature at 24°C. All the patients were dressed in standard gowns and leggings up to mid-thigh then covered by a standard blanket. Humidified oxygen (5 L min−1) was supplemented with nasal prongs. The distending medium used for hysteroscopy (0.9% NaCl) was warmed (37°C) in a standard fluid warmer. The total used volume of the distending medium was recorded.
The patients were observed for the occurrence of shivering starting after the intrathecal injection until the wearing off of the spinal anaesthetic. The shivering severity was graded as: grade 0 (no shivering), grade 1 (piloerection or peripheral cyanosis, but without visible muscle activity), grade 2 (visible muscle activity confined to one muscle group), grade 3 (visible muscle activity in more than one muscle group), or grade 4 (gross muscle activity involving the whole body) [10].
Any shivering attack with a score > 2 was treated by IV meperidine (25 mg). Hypotension, defined as a decrease of systolic blood pressure below 90 mm Hg or decrease of the MAP more than 20% from the baseline measurement, was treated with incremental boluses of IV ephedrine (6 mg). Bradycardia, defined as heart rate ≤ 60 beats min−1, was treated with IV atropine (0.5 mg).
The perioperative sedation state was recorded just before the intrathecal anaesthetic, intraoperatively every 15 minutes, at PACU admission and then every 30 minutes until 4 hours after admission to the PACU.
After the procedure the patient was shifted to the PACU which was conditioned as the operating theatre. The patients were covered with a standard blanket.
The primary outcome in the present study was the occurrence of shivering. Shivering incidence (number of intraoperative and postoperative attacks), time to occurrence after the intrathecal anaesthetic, severity of the attack at time of development and 5 minutes after meperidine administration and its response after meperidine administration (a decrease of shivering severity to grade 0 was reported as complete response, but incomplete response meant a decrease in shivering severity to a grade above 0), time between meperidine administration and the complete response were recorded.
The secondary outcomes were the cumulative dose of meperidine received, the time for the first postoperative analgesia request, and the perioperative complications (nausea and vomiting, hypertension or hypotension, bradycardia or tachycardia, dry mouth, dizziness, and desaturation).
It was reported that the median incidence of shivering after neuraxial anaesthesia is 55% [1]. To obtain a 20% reduction in the incidence of shivering in the study groups with a power of 90% and α = 0.9, a sample size of 25 patients in each group was required. We recruited 30 patients in each group in anticipation of any drop out or data loss.
The results were presented in a tabulated manner and analysed using IBM SPSS software package version 20.0. (Armonk, NY: IBM Corp). Qualitative data were described using number and percent. The Kolmogorov-Smirnov test was used to verify the normality of distribution. Quantitative data were described using range (minimum and maximum), mean, standard deviation and median. Significance of the obtained results was set at the 5% level. For categorical variables, the χ2 test was used to compare between different groups. Fisher’s exact or Monte Carlo correction for χ2 was used when more than 20% of the cells had an expected count less than 5. The F-test (ANOVA) was used for normally distributed quantitative variables, to compare between more than two groups, and the post hoc test (Tukey) for pairwise comparisons was used. For abnormally distributed quantitative variables, the Kruskal-Wallis test was used to compare between more than two studied groups and post hoc (Dunn’s multiple comparisons test) for pairwise comparisons.
RESULTS
The present study included 120 women who were randomized in equal numbers to receive oral premedication with tramadol, pregabalin, clonidine, or placebo (Figure 1). There were no significant differences between the studied groups in terms of demographic data or the volume of the distending medium (Table 1).
TABLE 1
Comparison between the four studied groups according to demographic data and volume of the distending medium
Regarding the primary outcomes of the present study, a significantly higher incidence of shivering (43.3%) was recorded in group O than the other groups (P = 0.025). Group C had the lowest incidence of shivering (13.3%), followed by group T (16.7%) with a statistically insignificant difference between them while being significantly lower than that in group P (20%). There was a statistically insignificant difference in the total number of perioperative shivering attacks among groups T, P and C (P > 0.05). None of group C patients developed more than 2 shivering attacks in comparison to 1, 3 and 4 patients in groups T, P, and O respectively (P = 0.004). The number of intraoperative attacks was significantly lower in premedication groups compared to the placebo group as well (P = 0.009), with insignificant differences between the premedication groups (P > 0.05). In the postoperative phase, a single shivering attack was recorded in 4 (13.3%), 5 (16.7%), 9 (30%) and 12 (40%) patients in groups C, T, P and O respectively (P = 0.069). The time to the first attack was significantly longer in group C (47.0 ± 5.7 minutes) than groups T, P and O (35.0 ± 3.5, 21.2 ± 4.9 and 16.0 ± 2.6 minutes respectively) (P < 0.001); and in group T than groups P and O as well (P < 0.001). The time to the second attack was significantly longer in group T than group O (P = 0.035) while being insignificantly different between groups T and P and P and O (P = 0.486 and P = 0.076 respectively). The median severity of the first attack was significantly higher in group O than other groups (P = 0.002), which in turn did not show any statistical significance between each other (P > 0.05). The severity of the first attack 5 minutes after meperidine treatment and that of the second attack both before and after meperidine treatment were insignificantly different between the 4 groups (P > 0.05). The shortest time until disappearance of the first attack was recorded in group C (1.1 ± 0.1 minutes); it was significantly shorter than that in groups O and P (1.6 ± 0.1 and 1.4 ± 0.1 minutes respectively) (P < 0.001) while being insignificantly different from group T (P = 0.546). Similarly, the time until disappearance of the first attack was significantly shorter in groups T and P than group O (P < 0.05). Furthermore, group T recorded the shortest time until disappearance of the second attack followed by groups P and O respectively with statistical significance between all of them (P < 0.05). The response of all the perioperative shivering attacks to meperidine treatment and the severity of the postoperative shivering attacks both before and after meperidine treatment were insignificantly different across the studied groups (P > 0.05). Ten percent of group C patients received meperidine, compared to 16.7%, 20% and 50% in groups T, P and O respectively (P < 0.001). The longest time for the first postoperative analgesia request was found in group P followed by groups C, T and O with statistical significance between all of them (P < 0.001) (Table 2).
TABLE 2
Comparison between the four studied groups according to shivering analysis
| Parameter | Group T (n = 30) | Group P (n = 30) | Group C (n = 30) | Group O (n = 30) | Test of significance | P |
|---|---|---|---|---|---|---|
| Shivering occurrence | 5a (16.7%) | 6a,b (20.0%) | 4a (13.3%) | 13b (43.3%) | χ2 = 9.317* | 0.025* |
| Total No. of shivering attacks | 0b (0–3) | 0b (0–3) | 0b (0–2) | 1a (0–3) | H = 14.574* | 0.002* |
| Significance between groups | P1 = 0.550, P2 = 0.729, P3 = 0.002*, P4 = 0.346, P5 = 0.012*, P6 = 0.001* | |||||
| 0 | 21a (70.0%) | 19a (63.3%) | 22a (73.3%) | 8b (26.7%) | χ2= 21.811* | 0.004* |
| 1 | 4a (13.3%) | 3a (10.0%) | 5a (16.7%) | 13b (43.3%) | ||
| 2 | 4a (13.3%) | 5a (16.7%) | 3a (10.0%) | 5a (16.7%) | ||
| 3 | 1a,b (3.3%) | 3a,b (10.0%) | 0b (0.0%) | 4a (13.3%) | ||
| No. of intraoperative attacks | 0 (0–2) | 0 (0–2) | 0 (0–1) | 1 (0–2) | H = 11.549* | 0.009* |
| Significance between groups | P1, = 0.564, P2 = 0.667, P3 = 0.007*, P4 = 0.314, P5 = 0.034*, P6 = 0.002* | |||||
| 0 | 22a (73.3%) | 20a (66.7%) | 23a (76.7%) | 12b (40.0%) | χ2 = 12.618* | 0.036* |
| 1 | 6a (20.0%) | 7a (23.3%) | 7a (23.3%) | 13a (43.3%) | ||
| 2 | 2a,b (6.7%) | 3a,b (10.0%) | 0b (0.0%) | 5a (16.7%) | ||
| No. of postoperative attacks | 0 (0–1) | 0 (0–1) | 0 (0–1) | 0 (0–1) | H = 7.022 | 0.071 |
| 0 | 25 (83.3%) | 21 (70%) | 26 (86.7%) | 18 (60.0%) | χ2 = 7.289 | 0.063 |
| 1 | 5 (16.7%) | 9 (30%) | 4 (13.3%) | 12 (40.0%) | ||
| Time of the first intraoperative attack (min) | 35.0 ± 3.5 | 21.2 ± 4.9 | 47.0 ± 5.7 | 16.0 ± 2.6 | F = 81.104* | < 0.001* |
| Significance between groups | P1 < 0.001*, P2 = 0.001*, P3 < 0.001*, P4 < 0.001*, P5 = 0.053, P6 < 0.001* | |||||
| Time of the second intraoperative attack | 53.5 ± 3.5 | 48.4 ± 5.8 | – | 38.7 ± 4.0 | F = 5.889* | 0.032* |
| Significance between groups | p1 = 0.486, p3 = 0.035*, p5 = 0.076 | |||||
| Severity of the first attack | 0 (0–4) | 0 (0–4) | 0 (0–3) | 1.5 (0–4) | H = 14.776* | 0.002* |
| Significance between groups | P1, = 0.680, P2 = 0.489, P3 = 0.004*, P4 = 0.269, P5 = 0.013*, P6 < 0.001* | |||||
| Severity of the second attack | 0 (0–3) | 0 (0–3) | 0 (0–1) | 0 (0–3) | H = 2.434 | 0.487 |
| Severity of the first attack 5 min after meperidine | 0 (0–1) | 0 (0–1) | 0 (0–0) | 0 (0–1) | H = 5.313 | 0.150 |
| Severity of the second attack 5 min after meperidine | 0 (0–0) | 0 (0–0) | 0 (0–0) | 0 (0–0) | H = 0.0 | 1.000 |
| Time until disappearance of the first attack (min) | 1.0 ± 0.1 | 1.4 ± 0.1 | 1.1 ± 0.1 | 1.6 ± 0.1 | F = 56.969* | < 0.001* |
| Significance between groups | P1 < 0.001*, P2 = 0.546, P3 < 0.001*, P4 = 0.006*, P5 = 0.016*, P6 < 0.001* | |||||
| Time until disappearance of the second attack (min) | 0.9 ± 0.1 | 1.2 ± 0.0 | – | 1.4 ± 0.1 | F = 33.250* | < 0.001* |
| Significance between groups | P1 = 0.005*, P3 < 0.001*, P5 = 0.034* | |||||
| Response of the first attack | ||||||
| Incomplete | 1 (20.0%) | 3 (50.0%) | 2 (50.0%) | 4 (36.4%) | χ2 = 1.461 | 0.802 |
| Complete | 4 (80.0%) | 3 (50.0%) | 2 (50.0%) | 7 (63.6%) | ||
| Response of the second attack | ||||||
| Incomplete | 0 (0.0%) | 0 (0.0%) | – | 0 (0.0%) | – | – |
| Complete | 1 (100.0%) | 3 (100.0%) | – | 5 (100.0%) | ||
| Shivering severity in the PACU | 0 (0–2) | 0 (0–2) | 0 (0–2) | 0 (0–4) | H = 3.547 | 0.315 |
| Severity of the PACU shivering attack 5 min after meperidine | 0 (0–1) | 0 (0–1) | 0 (0–0) | 0 (0–1) | H = 3.480 | 0.323 |
| Time to the first postoperative attack (min) | 71.0 ± 2.7 | 68.3 ± 5.5 | 81.8 ± 6.5 | 66.0 ± 2.8 | F = 12.404* | < 0.001* |
| Significance between groups | P1 = 0.688, P2 = 0.007*, P3 = 0.195, P4 < 0.001*, P5 = 0.610, P6 < 0.001* | |||||
| Response of the PACU attack | ||||||
| Incomplete | – | 4 (57.1%) | – | 7 (58.3%) | χ2 = 0.003 | 1.000 |
| Complete | – | 3 (42.9%) | – | 5 (41.7%) | ||
| Meperidine requirement | ||||||
| No | 25a (83.3%) | 24a (80.0%) | 27a (90.0%) | 15b (50.0%) | χ2 = 15.415* | 0.001* |
| Yes | 5a (16.7%) | 6a (20.0%) | 3a (10.0%) | 15b (50.0%) | ||
| Total meperidine consumption (mg) | 35.0 ± 13.7 | 37.5 ± 13.7 | 25.0 ± 0.0 | 33.3 ± 12.2 | F = 0.716 | 0.552 |
| Time for first postoperative analgesia request (min) | 187.2 ± 9.2 | 277.5 ± 8.8 | 209.5 ± 8.8 | 169.4 ± 5.7 | F = 985.65* | < 0.001* |
| Significance between groups | P1 < 0.001*, P2 < 0.001*, P3 < 0.001*, P4 < 0.001*, P5 < 0.001*, P6 < 0.001* | |||||
F – ANOVA test, Pairwise comparison between each 2 groups was done using post hoc test (Tukey). H – Kruskal Wallis test, Pairwise comparison bet. each 2 groups was done using post hoc test (Dunn’s for multiple comparisons test). χ2 – chi square test.
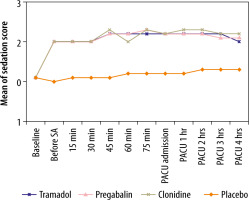
The sedation scores were significantly higher in groups T, C and P than group O before spinal anaesthesia (P < 0.001). This difference was sustained until the end of study’s follow-up with an average score of 2-2.3 in premedication groups (P < 0.001). However, no significant differences between premedication groups were observed at any time point (P > 0.05; Figure 2).
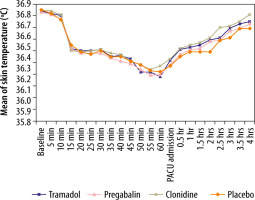
In terms of adverse events, the incidence of dizziness in group T was 33.3%, compared to 20%, 6.7% and 0% in groups P, C and O respectively (P < 0.001). Otherwise, the incidences of blood pressure and heart rate changes and perioperative nausea and vomiting were comparable between studied groups (P > 0.05) (Table 3). There was no single desaturation episode in any of the study participants. Despite the observed trend of a drop in body temperature, the changes in skin and tympanic membrane temperatures were not significantly different between the studied groups (P > 0.05; Figures 3 and 4).
TABLE 3
Comparison between the four studied groups according to side effects
| Parameter | Tramadol (n = 30) | Pregabalin (n = 30) | Clonidine (n = 30) | Placebo (n = 30) | χ2 | P | |
|---|---|---|---|---|---|---|---|
| Nausea and vomiting | |||||||
| Intraoperative | 2 (6.7%) | 1 (3.3%) | 1 (3.3%) | 0 (0.0%) | 2.062 | 0.905 | |
| Postoperative | 2 (6.7%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 3.599 | 0.244 | |
| MAP changes | Intraoperative | ||||||
| No change | 22 (73.3%) | 27 (90.0%) | 25 (83.3%) | 27 (90.0%) | 3.816 | 0.258 | |
| Hypotension | 8 (26.7%) | 3 (10.0%) | 5 (16.7%) | 3 (10.0%) | |||
| Hypertension | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | |||
| Postoperative | |||||||
| No change | 30 (100.0%) | 28 (93.3%) | 27 (90.0%) | 30 (100.0%) | 4.693 | 0.168 | |
| Hypotension | 0 (0.0%) | 2 (6.7%) | 3 (10.0%) | 0 (0.0%) | |||
| Hypertension | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | |||
| HR changes | Intraoperative | ||||||
| No change | 23 (76.7%) | 26 (86.7%) | 26 (86.7%) | 28 (93.3%) | 3.299 | 0.380 | |
| Tachycardia | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | |||
| Bradycardia | 7 (23.3%) | 4 (13.3%) | 4 (13.3%) | 2 (6.7%) | |||
| Postoperative | |||||||
| No change | 30 (100.0%) | 29 (96.7%) | 26 (86.7%) | 30 (100.0%) | 6.223 | 0.054 | |
| Tachycardia | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | |||
| Bradycardia | 0 (0.0%) | 1 (3.3%) | 4 (13.3%) | 0 (0.0%) | |||
| Dizziness | 10a (33.3%) | 6a,b (20.0%) | 2b,c (6.7%) | 0c (0.0%) | 15.547* | 0.001* | |
| Dry mouth | 1 (3.3%) | 2 (6.7%) | 4 (13.3%) | 0 (0.0%) | 4.602 | 0.207 | |
DISCUSSION
The results of this trial verified the efficacy and safety of oral premedication with clonidine, pregabalin, or tramadol in preventing post-spinal shivering. Clonidine, pregabalin, and tramadol effectively reduced the incidence, frequency, severity and duration of shivering attacks and perioperative meperidine consumption. Moreover, these medications significantly prolonged the time until the first shivering attack and first postoperative analgesic request. On the other hand, the findings showed that clonidine was more effective than tramadol and pregabalin in decreasing the severity, duration and number of post-spinal shivering attacks. Tramadol showed similar trends when compared to pregabalin. In terms of safety profile, tramadol and pregabalin appeared to increase the risk of dizziness compared to clonidine; however, no other major adverse events were observed.
The overall incidence of shivering in this study was 23.3% compared to the reported incidence of 40–70% in some other studies [2, 3]. This may be attributed to the prophylactic pharmacological and nonpharmacological measures used in this study. As any anti-shivering medication will decrease the metabolic heat generation [11], we combined the nonpharmacological measures to compensate for that. When non-pharmacological measures were not used in patients undergoing elective abdominal, orthopaedic and gynaecological surgery under spinal anaesthesia, shivering developed notably earlier than that recorded in our study [12].
Clonidine acts centrally and peripherally as a selective partial α2 adrenergic receptor agonist with sedative and analgesic properties [13]. Although it was previously thought that clonidine exerts an anti-shivering effect by peripherally inhibiting the sympathetic activity and vasoconstriction, a cumulative body of evidence demonstrated that clonidine also acts centrally by reducing the thermoregulatory thresholds for vasoconstriction and increasing the sweat thresholds [14, 15]. It also decreases spontaneous firing in the locus coeruleus, which is a pro-shivering centre. Therefore, a growing number of published trials assessed the prophylactic effects of clonidine on post-spinal shivering. In the present trial, we demonstrated that premedication oral clonidine was an effective prophylaxis against post-spinal shivering and it significantly reduced the incidence, severity, and duration of shivering attacks. In concordance with our findings, several double blinded, placebo-controlled trials reported that oral clonidine premedication significantly reduced the incidence, severity and duration of post-spinal shivering [16–18]. Furthermore, 2 meta-analyses showed a significant reduction in the incidence of post-spinal shivering after prophylactic oral or IV administration of clonidine [19, 20]. When lower doses (0.15 mg) of oral clonidine premedication were used before spinal anaesthesia for urological surgeries, despite showing significant anti-shivering properties, higher incidence, frequency and severity of shivering than those found in our study were recorded [13, 21].
On the other hand, meperidine, an agonist of κ and μ-opioid receptor, has been previously considered as the gold standard treatment option for post-anaesthesia shivering due to its potent inhibitory effects on the spinal pathway [6]. However, it was reported that meperidine was associated with a high rate of adverse events such as nausea, vomiting, and respiratory failure [22]. Tramadol, a synthetic opioid with weak activity on κ and μ-opioid receptors, inhibits the reuptake of 5-HT, norepinephrine, and dopamine and facilitates 5-HT release. Its anti-shivering effects may also be partially explained by its effects at the level of the pons. Thus, it has emerged as a more effective and safer alternative for prevention and management of post-anaesthetic shivering [23, 24]. Our trial showed that oral tramadol was effective in reducing the incidence, frequency, duration and severity of post-spinal shivering and meperidine consumption. In agreement with our results, tramadol was reported to be effective in decreasing the incidence and severity of shivering after spinal or general anaesthesia [5, 25–27]. Our study results showed that the efficacy of anti-shivering medications greatly varies between its prophylactic and therapeutic uses. After IV therapeutic use, tramadol was reported to have superior anti-shivering properties (a better response rate and shorter response time but with a comparable recurrence rate) than clonidine [28]. Gupta et al. [29] premedicated patients undergoing abdominal hysterectomy under spinal anaesthesia with oral clonidine (0.1 mg) or tramadol (50 mg). In comparison to our findings, they reported comparable shivering incidences (13.3% and 23.3%) but with earlier onset time (30 minutes). The comparable incidence despite the lower doses may be attributed to the older age of their participants and the different nature of the surgery. Tewari et al. [30] premedicated patients undergoing transurethral resection of the prostate with either clonidine (0.15 mg) or tramadol (50 mg) and reported lower incidences of shivering but with earlier onset time than our study results. Their lower shivering incidences may relate to the difference in age and gender of their population.
Pregabalin is an anticonvulsant that demonstrates efficacy in reducing neuropathic pain, anxiety, and fibromyalgia [31]. Owing to its regulatory effects on calcium influx through calcium channels in presynaptic neurons and gamma-aminobutyric acid (GABA) receptors, recent reports have proposed that pregabalin can be a promising prophylactic option for post-anaesthetic shivering, with conflicting results [7, 32]. For example, Ozgencil et al. [33] reported that pregabalin lowered the incidence of shivering compared to placebo after general anaesthesia for spine surgery. Meanwhile, Hashemian et al. [7] observed no significant impact of pregabalin on shivering incidence after general anaesthesia for laparoscopic cholecystectomy. This may be attributed to the use of pethidine for postoperative pain relief, which may have suppressed the shivering occurrence. However, no report, to the best of our knowledge, has assessed the prophylactic role of pregabalin after spinal anaesthesia. In the present trial, pregabalin exhibited significant efficacy in reducing the incidence and severity of post-spinal shivering.
The difference in shivering incidence despite the absence of significant difference in temperature changes among the groups reinforces the suggestion that mechanisms other than heat loss and subsequent drop in core temperature contribute to shivering pathogenesis. Another reinforcing factor is the improvement of shivering after meperidine despite the persistent trend towards hypothermia in all groups. The used drugs in our study have no thermogenic effects; therefore their anti-shivering properties may be related to lowering of the shivering threshold. Moreover, pain and temperature signals are conducted along similar pathways which synapse in dorsal horn regions [15]. It has been reported that pain may provoke shivering and that its alleviation may prevent non-thermoregulatory shivering [6, 34]. The analgesic properties of the drugs used in the current study may also have contributed.
We found, as did other studies, that clonidine was the most effective oral premedication option for reducing shivering incidence, frequency, severity, and duration while prolonging the analgesia duration, followed by tramadol then pregabalin [12, 30, 35].
The side effects profile recorded in our study is consistent with the findings of other reports that showed higher incidence of dizziness after tramadol or pregabalin premedication [36-39]. Interestingly, oral premedication with clonidine was not associated with hypotension or bradycardia, which are commonly reported after its IV use. The incidence of nausea and vomiting was higher after tramadol than clonidine, which is contrary to that reported after IV therapeutic use [12, 28]. We found, as did other investigators, a stable haemodynamic profile without a statistically significant difference among the study groups. This verifies the relatively better safety and tolerability of the oral prophylaxis than the IV treatment of shivering [29]. The 3 drugs were associated with significantly better sedation states than placebo premedicated patients.
We appreciate that the use of a fixed dose of the premedication agents is a limitation of the current study. However, this should not have affected the results considering the comparable body mass index of the participants. Further studies are warranted to determine the optimal dose of any of these drugs.
CONCLUSIONS
In conclusion, our results highlighted that clonidine was more effective and tolerable than tramadol and pregabalin for reduction of the incidence, severity, frequency and duration of post-spinal shivering as well as prolongation of the times until the first shivering attack and the first analgesic request in patients undergoing hysteroscopic procedures. This reinforces the concept that prophylaxis is better than treatment. The oral route showed a simple, low cost and readily available premedication form. Our findings have implications for the prevention and management of post-spinal shivering provided they are confirmed in large-scale trials.