Purpose
Carcinoma cervix is the second most common cancer in females and the third most common cancer in India [1]. The treatment modalities for cervical cancer are surgery, radiotherapy and chemotherapy depending on the stage and performance status of patients [2]. Radiotherapy plays an important role in the early and advanced stages of the disease [3]. For early-stage disease, radical radiotherapy constitutes a good option for medically inoperable patients [4]. For locally advanced intact cervical carcinoma patients (IN-CC), concurrent chemo-radiotherapy with weekly cisplatin followed by brachytherapy is the current standard of care [5-9]. Radical radiotherapy comprises external beam radiotherapy (EBRT) and brachytherapy (BT). EBRT (45-50.4 Gy) aims to reduce the gross tumor and control the microscopic disease in the pelvic area, while brachytherapy in the form of intracavitary radiotherapy (ICRT) is used to boost the primary tumor to a cumulative EQD2 (equivalent dose at 2 Gy per fraction) of 80 to 90 Gy [10]. In patients with distorted anatomy such as a narrow vaginal fornix, non-negotiable cervical os, persistent parametrial disease after EBRT, lower vaginal extension and unfavorable dosimetry with ICRT, high-dose-rate (HDR) interstitial brachytherapy (ISBT) or hybrid interstitial brachytherapy (HBT) is recommended [11]. In patients with vault carcinoma (VA-C), ISBT is indicated for lesions having a thickness of more than 5 mm, as it provides excellent target coverage with a minimum dose to the organs at risk.
In intracavitary brachytherapy, we primarily intend to treat the high-risk clinical target volume (HR-CTV), which includes the entire cervix, gross residual disease and positive parametrial disease. By virtue of lateral throw-off of isodose distribution obturator lymph nodes receive an incidental 20-25% dose by ICRT [12]. In general, the dose to pelvic lymph nodes from ICRT is recorded at point B (which is located at the level of point A and 5 cm from the midline) and at the different reference points of the lymphatic trapezoid [13]. The dose to the obturator group of lymph nodes is presumed to be represented by point B.
Although studies have assessed doses to pelvic lymph nodes (PLN) in ICRT [14, 15], the same findings may not be extrapolated to the ISBT treatments as the geometry of the applicators are different and by the virtue of multiple straight and divergent needles with or without central tandem, the doses to PLN from ISBT may be very different from traditional ICRT applications. Because of the geometry of the application, different pelvic nodal groups may receive different doses. There is a paucity of data evaluating the doses received by PLN by ISBT.
Hence, we intended to prospectively evaluate the doses to PLN groups in IN-CC and VA-C patients treated with ISBT.
Material and methods
Ten cervical carcinoma patients (6 patients with IN-CC, 4 with VA-C) were recruited for this prospective dosimetric study between January 2018 and June 2020. Patients with indications of ISBT for either IN-CC or VA-C, ages 18 to 65 years, with a Karnofsky performance status (KPS) ≥ 70, hemoglobin ≥ 10 g/dl, total leukocyte count ≥ 4000/mm, platelet count ≥ 100,000/mm3, creatinine clearance ≥ 60 ml/min, and normal liver function test were included in this study. Patients with non-squamous histology, para-aortic lymph nodes, distant metastases, or synchronous/metachronous malignancy were excluded. Detailed medical history, complete gynecologic examination, complete blood count, liver and kidney function tests, chest X-ray and contrast-enhanced magnetic resonance imaging (MRI) of the abdomen and pelvis at baseline and after completion of EBRT were included in pretreatment work-up.
After obtaining written informed consent as per the institutional protocol, all eligible patients received EBRT to the whole pelvis delivered with three-dimensional conformal radiotherapy technique with a total prescription dose of 45 to 50.4 Gy in 25-28 fractions over 5 to 5.5 weeks with concurrent cisplatin administered intravenously once a week at a dose of 40 mg/m2. Based on clinical examination and MRI of the pelvis after completion of EBRT, those not found suitable candidates of ICRT or having carcinoma vault received ISBT. Brachytherapy was started within a week of completion of EBRT.
Brachytherapy implant procedure
The brachytherapy implant procedure was performed under spinal anesthesia by using a MUPIT (Martinez universal perineal interstitial template) in the lithotomy position. Foley’s tri-way urinary catheter insertion was done. Afterward per vaginal and per rectum examination was done to assess the extent of disease. Under the trans-rectal ultrasonography guidance (TRUS) probe, the rectum and bladder were identified first by visualizing Foley’s bulb and the orientation of other pelvic structures was determined. A uterine sound was used to check the patency of the cervical os in IN-CC patients and a central tandem was introduced in the uterine cavity. The MUPIT obturator was moved into the vagina until it fit against the cervix. In the VA-C patient, the obturator was inserted into the vagina and the inner plate of the template was fitted over it and fixed with a screw. The plate was positioned against the wall of the perineum and stitched to the skin. Stainless steel needles 20 cm in length were inserted one by one through the perineum under TRUS guidance. The divergent needles were implanted to cover the lateral extent of disease based on the clinical and radiological findings. After inserting the desired number of needles, a digital rectal examination was done to rule out rectal injury by needles and the needles were fixed with screws. Outer and inner perineal plates were fixed by ensuring alignment.
Treatment planning of ISBT
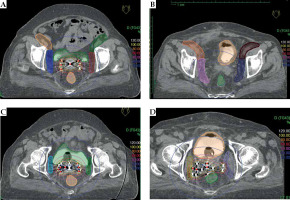
A computed tomography (CT) scan of the whole pelvis was done with slice thickness of 3 mm. Intravenous contrast was not given. Brachytherapy planning was performed on the Oncentra treatment planning system version 4.5.3 (Nucletron, an Elekta company, Elekta AB, Stockholm, Sweden) where the target, organ at risk and PLN were contoured. The extent of the target was decided by using the length of needles having clinical and radiological findings in mind. The target was drawn out by the line connecting the needles that were topmost on each CT slice. External iliac (EI-N), internal iliac (II-N), obturator (OB-N) and sacral (SA-N) groups of lymph nodes were delineated (Figure 1) by using the Taylor et al. guidelines [16]. Briefly, the external iliac nodes were drawn by 7 mm expansion (except anteriorly where it was 17 mm) along the external iliac blood vessel. The internal iliac nodes were drawn by providing a 7 mm margin around internal iliac blood vessels. Obturator nodes were drawn by joining external and internal lymph nodes with the width of 18 mm wide strips along the pelvic wall. Pre-sacral nodes were drawn by providing a 10 mm strip over S1-S3. Needles were reconstructed and a step size of 2.5 mm was used. The dose was prescribed to the basal point as per ICRU 58 recommendations [17]. Only dwell positions present within the target volume were activated. The central tandem was loaded from the tip until the level of the flange (which was at the top of the obturator) with loading at alternate dwell positions and were activated with similar dwell weights as the needles to begin with. Afterwards, using the manual optimization method, dwell times of the central tandem were increased to the simulated dwell time of the central tandem with the same dose prescription on a standard ICRT plan of the same day to replicate the point A lateral dose throw off from the tandem contribution. Manual optimization was again used to control excessive dose heterogeneity by balancing the loading of different dwell positions adjacent to the central tandem to achieve the desired dosimetry. Both graphic and geometric optimization was done to achieve the best plan. A dose of 10 Gy was prescribed to the target in IN-CC patients and 8 Gy in VA-C patients. Each patient had two ISBT implants one week apart (total 20 sessions of ISBT). Doses received by 100%, 90% and 50% volume (D100, D90, D50) and 2 cm3, 1 cm3 and 0.1 cm3 volumes (D2cc, D1cc, D0.1cc) in percentage of the prescribed doses were evaluated for each nodal group. After the approval of plan, patients were transferred to the brachytherapy suite. Brachytherapy was delivered with an iridium-192 source (Microselectron, high-dose rate [HDR], Nucletron, an Elekta company, Elekta AB, Stockholm, Sweden) for all patients. Implant needles were taken out after completion of the treatment with post-operative care and patients were discharged the next day.
Statistical analysis
All data were analyzed by the SPSS statistical software package for Mac (Version 25.0; IBM, Armonk, NY, USA). Quantitative variables were described in terms of mean with standard deviation. Doses to lymph nodes in patients with a central tandem (IN-CC) were compared to those without a central tandem (VA-C patients) using Student’s t-test and a p-value ≤ 0.05 was considered significant.
Results
Twenty ISBT procedures were performed in 10 patients. Patient characteristics are described in Table 1. Stage distribution for IN-CC patients was stage IIB : IIIB : IIIC1 in 2 : 3 : 1 patient respectively as per the FIGO 2018 staging system (International Federation of Gynecology and Obstetrics). Persistent parametrial disease was present in 3 of the patients after EBRT. The pelvic lymph node in one patient with IIIC1 disease responded well to EBRT and hence none of the patients had positive pelvic LN at the time of ISBT. Of 4 VA-C patients, 2 had parametrial infiltration (with the parametrium remaining persistent after EBRT in one case). None of the VA-C patients had lymph node positive disease.
Table 1
Summary of patient characteristics
Median number of needles was 18 (range, 16-20). Median number of divergent needles was 4 (range, 4-8). Mean target volume for IN-CC and VA-C patients was 80.2 ±4.33 cc and 66 ±5.1 cc respectively. Mean height, width and thickness of the target were 7.8 ±1.1 cm vs. 4.8 ±1.2 cm, 6.4 ±0.8 cm vs. 6.5 ±1.3 cm and 2.6 ±0.6 cm vs. 2.4 ±0.7 cm in IN-CC vs. VA-C patients respectively.
Mean target V95 (volume receiving 95% of the prescribed dose) was 76 cc (range, 70-84 cc) for IN-CC patients and 62 cc (range, 54-65) for VA-C patients. The median total reference air-kerma (TRAK) values for IN-CC and VA-C were 0.64 (0.55-0.69) and 0.37 (0.33-0.45), respectively, and this has been updated in the revised manuscript.
Mean D2cc to bladder per fraction was 6.2 Gy (range, 5.8-7.1) for IN-CC patients and 4.8 Gy (range, 3.8-5.2 Gy) for VA-C patients. Similarly, mean D2cc to rectum per fraction was 6.3 Gy (range, 5.1-6.8 Gy) for IN-CC patients and 4.5 Gy (range, 3.6-5.3 Gy) for VA-C patients.
Doses received by the respective nodal groups of the right/left side and combined doses (right + left) for the whole cohort of IN-CC and VA-C patients are summarized in Table 2. Patients with a central tandem (IN-CC) received significantly higher doses to external, internal iliac and sacral groups of lymph nodes (all p < 0.001 except D100 of EI-N p = 0.004) as compared to those without a central tandem (VA-C). Doses to the combined bilateral OB-N D100 (p = 0.143), D90 (p = 0.078) and D50 (p = 0.133) were not statistically different in IN-CC patients versus VA-C. However, dose to combined bilateral OB-N D2cc (p = 0.003), D1cc (p = 0.007) and D0.1cc (p = 0.005) were significantly higher in IN-CC patients (with a central tandem) vs. VA-C (without a central tandem), as illustrated in Table 3.
Table 2
Doses received by respective nodal groups of right/left side and combined (right + left) for the entire cohort of patients in terms of percentage of prescribed dose
Table 3
Combined dose received by respective nodal groups with and without central tandem in terms of percentage of prescribed doses
Discussion
The presence of pelvic lymph nodes is an independent prognostic factor in carcinoma of the cervix [18]. Pelvic lymph node positivity for stages IIA, IIB, IIIA, and IIIB/IV has been reported to range between 10-45%, 26-62%, 39-59%, and 39-88% respectively [19-22]. Most of the pelvic nodes respond to EBRT with concurrent chemotherapy, but there is a need to boost the positive pelvic lymph node to a higher dose, which also depends on the size of the lymph node [23]. Nodal boost may be carried out with EBRT either by simultaneous integrated boost (SIB) or sequential boost. While SIB has the advantage of reduction of overall treatment time and total dose [24], it may lead to increased radiation-related morbidity when there are larger lymph nodes (> 3 cm). A sequential boost permits the adaptation based on nodal disease regression after EBRT and caters to the explicit calculation of the dose contribution from brachytherapy [25]. The contribution of the dose from BT ranges from 6% to 34% for obturator, external and internal iliac lymph nodes and is negligible at the common iliac nodes for ICRT [26]. Dose heterogeneity can occur close to the BT target if pelvic nodes are treated and therefore the cumulative dose to pelvic lymph nodes for EBRT and BT must be taken into consideration in the EQD2 calculations at the time of BT. Higher doses to pelvic nodal groups may also be counterproductive in leading to higher incidence of long-term morbidities such as lymphedema and insufficiency fractures.
Historically, doses received by the PLN during ICRT were estimated by the lymphatic trapezoid of Fletcher, which represented the dose to the lower para-aortic, lower common iliac and middle external iliac lymph nodes, and by the pelvic wall points, which denote the dose to the obturator lymph nodes [13]. Few studies have evaluated the dosimetry of the different pelvic lymph nodal groups with ICRT applications [14, 15, 27]. It has been concluded that there is a significant dose supplement to the PLN during ICRT. However, the extent of association between point B dose and dosimetric parameters of the individual groups of nodes is low. Therefore, it is important to determine the dose received by individual nodal groups during brachytherapy, at least in patients with enlarged lymph nodes, to calculate the cumulative dose delivered [26].
In those patients for whom ICRT is not feasible, ISBT is a valuable tool in both IN-CC and VA-C patients. The incidental doses to PLN from ISBT are still not known. In our study, we found that the mean D90 values of the combined bilateral OB-N, II-N, EI-N and SA-N groups were 33.62%, 6.98%, 5.1% and 7.76% of the prescribed doses respectively and mean D2cc values of the combined bilateral OB-N, II-N, EI-N and SA-N groups were 102.94%, 39.69%, 15.4% and 15.36% of the prescribed doses respectively.
The doses to the OB-N groups were higher in our study with ISBT as compared to OB-N doses from ICRT reported by Lee et al. [12]; D90 and D2cc were 38.59 ±4.74% vs. 17.3 ±3.6% and 121.48 ±4.94% vs. 27.4 ±4.7% respectively. While D90 to OB-N from ICRT reported by Rangarajan et al. [27] was comparable to our study (38.5 ±10% vs. 38.59 ±4.74% respectively), D2cc to OB-N was lower compared to our study (58.5 ±18% vs. 121.48 ±4.94% respectively). Owing to proximity of the OB-N to the interstitial needles, this group receives a very high dose from the ISBT and hence it would be prudent to take this into consideration while planning to boost positive lymph nodes in the OB-N region.
The doses to II-N group D90 were lower in our study as compared to those reported by Lee et al. [12] and Rangarajan et al. [27], as illustrated in Table 4; the doses to II-N D2cc were higher as compared to both these studies. The region of II-N in proximity to the needles will have a higher dose (D2cc) than the average dose received by the entire II-N group (D90), and hence the location of the positive II-N is important to predict the incidental doses to that region, and this must be individualized on a patient-to-patient basis.
Table 4
Comparison of combined dose received by respective nodal groups between our study and other reported studies in literature
| Parameter | Our study | Study by Lee et al. [12] | Study by Rangarajan et al. [27] | ||
|---|---|---|---|---|---|
| Bilateral combined mean dose without central tandem (%) | Bilateral combined mean dose with central tandem (%) | Bilateral combined mean dose (%) | Bilateral combined mean dose (%) | ||
| Obturator nodes | |||||
| D100 | 16.95 ±2.79 | 24.44 ±3.51 | 12.3 ±3.2 | 28.57 ±5.25 | |
| D90 | 26.15 ±3.91 | 38.59 ±4.74 | 17.3 ±3.6 | 38.5 ±10 | |
| D50 | 52.63 ±14.91 | 74.78 ±6.66 | 23.4 ±4.1 | 54 ±16 | |
| D2cc | 75.11 ±23.18 | 121.48 ±4.94 | 27.4 ±4.7 | 58.5 ±18 | |
| D1cc | 95.27 ±26.5 | 165.84 ±7.79 | 30 ±5.3 | 71.4 ±22.8 | |
| D0.1cc | 162.30 ±32.48 | 259.42 ±12.80 | 35 ±6.6 | 97.14 ±45.7 | |
| Internal iliac nodes | |||||
| D100 | 1.34 ±0.33 | 3.64 ±0.20 | 13.1 ±3.04 | 14 ±4.2 | |
| D90 | 3.94 ±0.46 | 9.01 ±0.46 | 17.5 ±3.4 | 20.1 ±6.1 | |
| D50 | 9.79 ±1.02 | 19.69 ±0.75 | 22.6 ±4.1 | 12.67 ±6.2 | |
| D2cc | 22.95 ±3.11 | 50.84 ±2.22 | 28.5 ±5.5 | 30 ±10.7 | |
| D1cc | 26.08 ±3.88 | 62.21 ±2.24 | 30.8 ±6.09 | 32.8 ±9.7 | |
| D0.1cc | 34.90 ±6.34 | 107 ±5.6 | 36 ±7.4 | 40 ±12.8 | |
| External iliac nodes | |||||
| D100 | 0.84 ±0.29 | 2.37 ±0.32 | 4.1 ±1.5 | 9.4 ± 3.14 | |
| D90 | 3.06 ±0.53 | 6.45 ±0.46 | 8.7 ±2.04 | 16.71 ±2.71 | |
| D50 | 6.49 ±0.56 | 10.10 ±0.55 | 14.6 ±3.23 | 22.7 ±4.2 | |
| D2cc | 8.67 ±0.74 | 14.35 ±0.49 | 20.5 ±4.5 | 30 ±5.8 | |
| D1cc | 11.98 ±1.13 | 19.38 ±0.67 | 22.4 ±4.7 | 34.2 ±6.7 | |
| D0.1cc | 15.99 ±0.81 | 22.18 ±0.86 | 26.6 ±6.6 | 40 ±7.4 | |
The brachytherapy dose contribution to the EI-N decreases considerably as compared to II-N and OB-N groups. D90 and D2cc to EI-N were comparable (albeit a bit lower) in our study as compared to those reported by Lee et al. [12] (Table 4). The lower doses in our study may be attributed to the different contouring guidelines used in studies. We used the Taylor et al. [16] guidelines, which mandate a 17 mm anterior extension for EI-N groups, and hence our volumes were larger and more lateral. Nevertheless, commensurate with other studies, we found that the EI-N receives approximately 6-10% of the prescribed doses, and hence nodes in these regions would need an EBRT boost most often.
In our study, VA-C patients received significantly lower mean D90 and D2cc to the combined bilateral II-N, EI-N and SA-N than in IN-CC patients (Table 3), and this is mainly because of higher contribution to PLN from the active sources in the central tandem. Although different dose prescription schedules may impact the doses received by the pelvic LN groups in absolute numbers, since we have reported all doses in terms of the percentage of the prescribed dose (Tables 2-3), this is unlikely to differ with different dose prescriptions. The results of our study would give an average idea of the doses received by the different pelvic LN groups electively. The dose received by an involved pelvic LN would certainly depend on the EBRT schedule, location, size of the LN, etc., and this must be calculated in a case-to-case basis as per the ICRU 89 recommendations. Other factors which may influence the doses to the PLN groups are optimization of the plan, type of applicator used (tandem ring vs tandem ovoid), normalization points etc. [12]. With the growing popularity of the hybrid applicators with the selective use of interstitial needles in addition to the standard ICRT applicators, the doses could be differentially impacted on the side of the additional needles. There are no specific studies addressing this issue, and it could be interesting to know the dose contribution to different PLN groups from these hybrid applicators.
To our best knowledge, none of the studies have evaluated and reported the incidental dose to PLN groups from pure HDR image-guided ISBT. Our study may help to understand the approximate dose to various PLN groups received by ISBT, which may be useful in planning nodal boost doses and techniques in the management of IN-CC and VA-C patients. One of the limitations of our study is the small number of patients, which may have resulted in some heterogeneity in dosimetric data. Future studies evaluating doses to positive lymph nodes at different pelvic nodal group locations during ISBT may help us to understand the dosimetric profile of incidental doses to involved pelvic lymph node stations.
Conclusions
In cervical carcinoma patients treated with HDR image-guided ISBT, pelvic lymph node groups receive significant doses, particularly obturator and internal iliac groups. The dose contribution to pelvic lymph nodes is higher in patients with intact cervical cancer where a central tandem is used as compared to post-operative patients.