Purpose
High-dose-rate interstitial brachytherapy (HDR-ISBT) is widely used for the treatment of pelvic tumors, including prostate cancer [1,2,3,4,5], uterine cervical cancer [6,7], recurrent cancer in pelvic area, and anorectal cancer [8,9,10,11,12].
Because HDR-ISBT requires applicator insertion, pelvic tumor treatment poses risks of bleeding, infection, or intestinal perforation. However, there are a few reports on complications caused by applicator implantation in HDR-ISBT [13]. Herein, we describe a case with recurrent bladder cancer in the pelvis who developed small bowel perforation during HDR-ISBT. To the best of our knowledge, this is the first report of small bowel perforation caused by applicator implantation in HDR-ISBT.
Case presentation
An 82-year-old man was diagnosed with recurrence of bladder cancer. He underwent radical cystectomy and ureterocutaneostomy for bladder cancer in 2007. Unfortunately, during the surgery, a rectal damage occurred, and he had to undergo temporal colostomy and multiple subsequent abdominal open surgeries for restoration. In 2015, a recurrent tumor (40 × 42 × 26 mm) was detected on computed tomography (CT) on the right side of the rectum (Figure 1A, B). The tumor was strongly suspected to be directly infiltrating the rectum, but infiltration to the surrounding small bowel was not evident. Contrast-enhanced total body CT detected no other tumors except the abovementioned lesion. The patient refused chemotherapy and surgery because of his age. He also refused external beam radiotherapy, which required long treatment period. He eventually agreed to undergo ISBT with radical intention because of its short treatment period and high local effect.
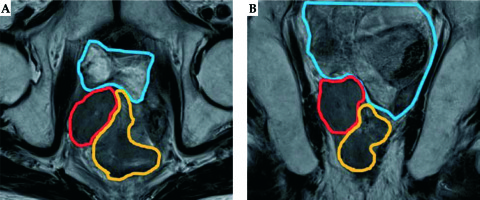
Fig. 1
Magnetic resonance image of the recurrent tumor. A) Axial image; B) Coronal image. The red line depicts the tumor, the orange line depicts the rectum, and the blue line depicts the small bowel
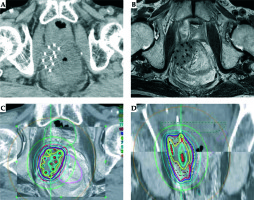
After administration of epidural anesthesia, 11 applicators (ProGuide Sharp Needle; Nucletron, ELEKTA AB, Stockholm, Sweden) were percutaneously inserted into the tumor under transrectal ultrasound (TRUS) guidance (ALOKA ProSound Alpha 7, Hitachi ALOKA Medical, Tokyo, Japan). Applicator insertion was completed without any trouble. Subsequently, we performed CT and magnetic resonance (MR) image, and radiation therapy was planned (Figure 2A, B). In the treatment planning system (Oncentra® brachy; Nucletron, ELEKTA AB, Stockholm, Sweden), we delineated the gross tumor volume (GTV) using the planning system function of CT/MR image registration (Figure 2C, D), and 6 Gy per fractionated dose was prescribed for 98% of the GTV. Minimum doses to the most exposed 2.0 cc (D2cc) of the rectum and the small bowel were 4.3 Gy and 4 Gy per fraction, respectively. HDR-ISBT of 54 Gy was planned to be administered in nine fractions for five days (twice daily with six hours interval), using a 192Ir remote after-loading system (MicroSelectron v3® HDR, Nucletron, ELEKTA AB, Stockholm, Sweden).
Fig. 2
Treatment planning. A) Computed tomography image; B) Magnetic resonance image; C) Treatment planning (axial image); D) Treatment planning (coronal image)
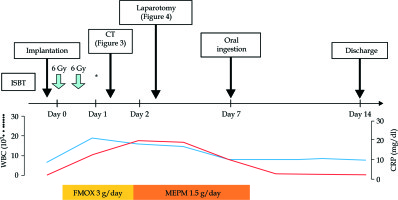
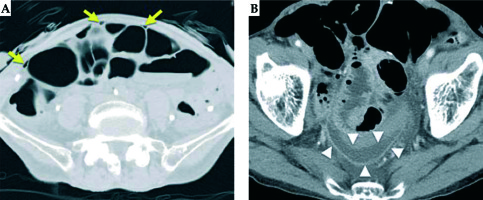
HDR-ISBT was started as planned. The patient had no clinical symptoms during or after the initial treatment. On the second day of treatment, however, his laboratory data showed an increased white blood cells count (19,200/μl) and C-reactive protein level (10.5 mg/dl) after two fractions of treatment and the total dose of 12 Gy. Although the patient had no clinical symptoms, we performed CT to detect any adverse events. Free air and a fluid collection surrounded by an enhanced ring were noted and imaging diagnosis of an abscess at pelvic floor was made (Figure 3A, B); however, we could not identify the exact location of intestinal perforation. We stopped the treatment and removed the applicators. Fasting management was started and the antibiotic treatment was changed from flomoxef (3 g/day) to meropenem (1.5 g/day). The following day, signs of local peritoneal irritation were noted and we decided to perform laparotomy.
Fig. 3
Computed tomography images one day after treatment initiation. A) The yellow arrows depict free air; B) The white arrowheads depict an abscess (a fluid collection surrounded by an enhanced ring)
Intraoperatively, a small amount of bleeding and pus was observed at the pelvic floor (Figure 4A) and the tumor adhered to the small intestine. Even then we could not directly pinpoint the perforation site. After careful irrigation of the abdominal cavity, a part of the small bowel that was attached to the tumor was removed.
Fig. 4
Perioperative imaging. A) Intraoperative image. The white arrow depicts the pus, the white arrowhead depicts hemorrhage; B) Resected small bowel after surgery. The white arrowhead depicts the applicator pathway; C) Resected small bowel after formalin fixation. The yellow dotted line depicts the tumor; D) Resected small bowel after formalin fixation (larger scale). The red line depicts the tumor and the yellow line depicts the mucosal surface
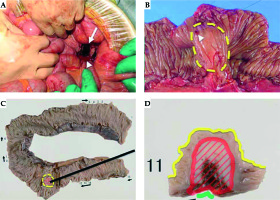
When we cut opened the resected small bowel, we recognized the site where the applicator was inserted into a part of the tumor on the mucosal surface (Figure 4B). Finally, pathological examination confirmed that the tumor had infiltrated the small intestine directly and that the infiltrated part had reached the submucosa (Figure 4C, D).
The postoperative course was uneventful. The patient started oral ingestion a week later and was discharged from the hospital two weeks after admission (Figure 5). He was followed up without further treatment, and there had been no tumor growth nor any other complications. The patient died 20 months later of systemic metastases from his bladder cancer.
Discussion
The method of applicator insertion under TRUS guidance has been very popular in the treatment of prostate cancers [1,2,3,4,5]. After Stock et al. first reported on the treatment of cervical cancer [14], the same method has been widely used in gynecological malignancies and some other recurrent tumors in the pelvic cavity [6,7,8,9,10,12,13].
ISBT for pelvic site tumors poses risks of bleeding, infection, and intestinal perforation. However, there has been only one report of complications caused by applicator implantation by Sharma et al. [13]. They also used TRUS guidance and observed a perioperative complication rate of 2.5% in the form of hematuria. They investigated the locations of the applicators using CT images obtained for treatment planning and they did not encounter any case with perforation. Thus, they suggested that TRUS assists in avoiding the needle injury/perforation of the pelvic structures, thereby reducing the risk of perioperative complications.
In our hospital, we have treated 82 patients who had tumors at pelvic sites with HDR-ISBT under TRUS guidance between September 2014 and August 2019. We retrospectively investigated whether we have inserted any of the applicators directly into the intestine, using MR images obtained for treatment planning. We had inserted a total of 1,014 applicators, and there were only two (0.2%) that appeared to be inserted into the intestine. Both applicators were observed in the patient of this report. None of the other patients had tumor clearly suspected of intestinal infiltration on MR images.
As mentioned above, MR images confirmed that two applicators were inserted into small intestine. In actual, however, only one applicator was found to penetrate the small intestine. The intestinal perforation might have occurred because the intestine penetrated by one applicator had an abnormal mucosa infiltrated directly by the tumor, while the intestine penetrated by another applicator had a normal mucosa, so the perforation in the second case might have not occurred.
The small bowel perforation may have been caused by mucosal insertion and mucosal irradiation. Because the perforation was observed soon after the start of BT, the treatment plan was made so that the dose to small bowel was reduced as much as possible, since the influence of mucosal insertion was considered significant.
In most c ases of postoperative tumor recurrence in the pelvis, there are intestines at the cranial side of tumors. If there is any possibility that the tumor had infiltrated the intestine, we recommend laparoscopy to be used [15,16,17].